|
12/24/2023 0 Comments Sigma bond hybridization 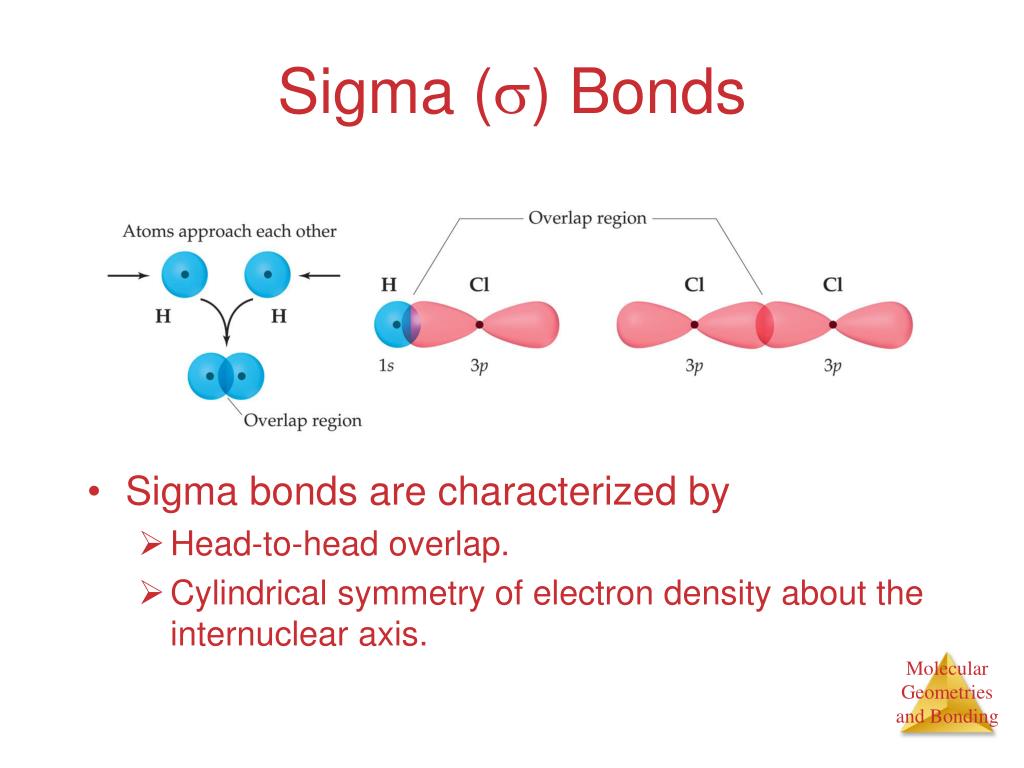
It also takes care of the steric number that is the number of regions of electron density surrounding the atom. Mainly, the VSEPR model focuses on the electron pairs around the central atoms. As discussed above, N2 forms a triple covalent bond and sp hybridization.Īs mentioned above, the Lewis structure only tells about which atoms have lone pairs but, valence-shell, electron-pair repulsion(VESPER) predicts the shape of many molecules. To understand the molecular geometry of any molecule, learning its Lewis structure and hybridization is very important. The next head-to-head overlapping of p-orbitals each containing one electron gives one more π bond.įrom the above explanation of overlapping, you can conclude that a single bond, double bond, and triple bond corresponds to a σ bond, σ bond plus a π bond, and a σ bond plus two π bonds respectively. On the other side, the two p-orbitals on both the atoms each containing one electron give a π bond. Sp hybridization includes overlapping of sp-orbitals on both the nitrogen atoms to form a σ bond. The setup results in N2 forming sp hybridization. A pi bond is made due to the presence of a second or third bond.įor nitrogen atom, the valence-shell electron configuration is 2s2 2px1 2py1 2pz1 where it shows that 1s and 1p orbitals are hybridizing to give a new set of two sp-orbitals.Sigma bond is the first bond that is made with other atoms. 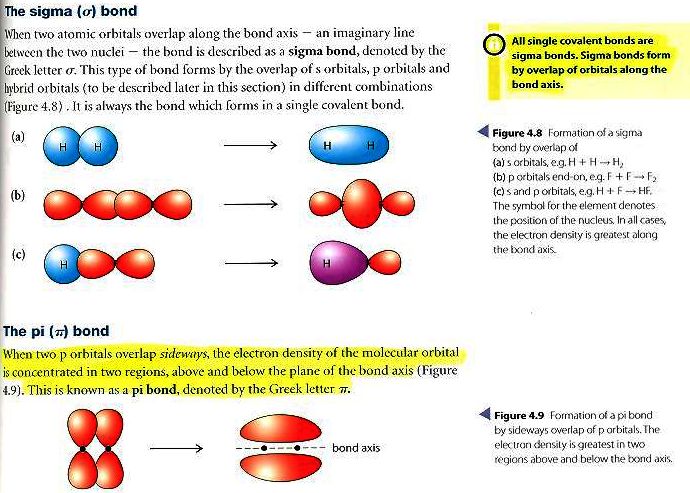
Both the bonds help to identify the type of hybridization by either forming head-to-head overlap or when 2p orbitals overlap. There are two types of bonds which are widely used in Chemistry, sigma (σ) and pi (π) bonds. Finally, after sharing three pairs of electrons that make the distribution of 6 electrons in a bond, it is known as a triple covalent bond.As per the octet rule, still each atom needs two more electrons to complete its outermost shell. After creating a single bond between the atoms, both atoms have 6 electrons each.6 electrons to make the correct structure. To follow the octet rule (eight electrons per atom), each Nitrogen atom needs 3 more electrons i.e.Show the remaining 3 electrons at the external side of each atom.Now, set up the covalent bond by writing both the Nitrogen atoms next to each other and draw a line to represent the bond.Take care of bonding and non-bonding electron pairs that directly influence the geometry of the Lewis structure.Both the atoms have the same electronegativity, there will be no central atom in the structure.Since you have 2 atoms of Nitrogen, assign the valence electrons using dots in a diagram to each atom-like 5 dots around each atom.Now, distribute valence electrons around the atoms of N2.Thus, 10 valence electrons need to be arranged in the structure to show the chemical bonding between two atoms of the Nitrogen molecule.The total number of electrons present in the valence shell is 5 * 2 = 10e. As per the molecule N2, it has two atoms of Nitrogen.2,5, it has five electrons in its outermost valence shell. Thus, as per the electronic configuration of the element i.e. In the Periodic Table, Nitrogen is placed in Group 5 across Period 2.Note: The most important thing about the Lewis dot structure is that only valence electrons take part in chemical bonding.īelow is the electron dot structure for a Nitrogen molecule: Arrange the remaining electrons to the terminal atoms.Choose the central atom by identifying the least electronegative atom.While representing the bonds, you should know about lone and bonded pairs.Take care of the octet rule where the ions or atoms should have eight electrons in their outermost valence shell (Duplet Rule: There is an exception in the case of Hydrogen that needs only two electrons to gain stability.).Calculate the total number of valence electrons of the atoms present in a molecule.Firstly, check out the atomic number of each atom from the Periodic Table.Lewis used lines to state a covalent bond between two electrons and each electron is denoted by a dot in the diagram. To determine the number of valence electrons, you can simply note down the Group number of the element from the Periodic Table. 
An electron that is placed in the outermost shell of an atom is known as a valence electron. Mainly, the structure depicts the arrangement of the valence shell electrons of an element. It also describes the chemical bonding between atoms present in the molecule. The Lewis structure indicates the atom and its position in the model of the molecule using its chemical symbol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
